Thomas Lectka and co-workers from Johns Hopkins University have reported in Chemical Science on an interesting C-C bond cleavage process.
Archive for the ‘C-C Bond Breaking’ Category
This reaction breaks C-C bonds.
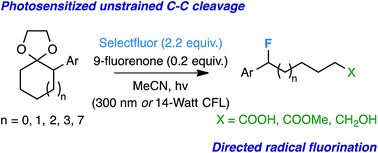
C-C bond cleavage of alpha-aryl acetals
Posted by naturalproductman on July 13, 2015
Posted in C-C Bond Breaking, Cascade Reactions, Light Mediated, Methodology | Leave a Comment »
C-C bond breaking mechanism
Posted by naturalproductman on November 8, 2012
Here’s an interesting paper where the authors used 18O2 to label one of the oxygens found in the product to investigate the mechanism. Now I’m going to be a little bit critical here but in the supporting information they put the 18O-label on the carbonyl oxygen but in the manuscript they don’t know if it is on the O-benzyl of the ester or the carbonyl oxygen. Moreover the abstract figure shows that the 18O is on the carbonyl oxygen but they really don’t know. It’s a bit deceiving in the paper because it kind of sounds like they determined it through GC-MS but in the supporting information I could not find the GC-MS trace. I guess there is a limitation in analytical technology to determine the position of the 18O. But still it is not clear from the paper that they don’t know which position the 18-oxygen is.

Posted in C-C Bond Breaking, Cascade Reactions, Mechanistic, Methodology | Leave a Comment »
C-C bond cleavage via SnCl4
Posted by naturalproductman on December 2, 2011
Matthias Brewer and co-workers at the University of Vermont have reported in Organic Letters on a tin promoted C-C bond cleavage.

Posted in C-C Bond Breaking, Cascade Reactions, Methodology, Ring expansion, Ring Opening | Leave a Comment »
Poof be gone
Posted by naturalproductman on September 18, 2011
Alexander Butin and co-workers have reported in Tetrahedron Letters on the loss of a vinyl methyl ketone group from an indole.

Posted in C-C Bond Breaking, Cascade Reactions, Methodology | Leave a Comment »