Archive for the ‘Zinc’ Category
Posted by naturalproductman on April 14, 2016
Xiaoming Feng and co-workers from Sichuan University have reported in ACIE on a Zn-catalyzed [2+2] reaction between alkynones and cyclic silyl enol ethers.
ACIE paper
Posted in Asymmetric, Cascade Reactions, Methodology, Transition Metal, Zinc, [2+2] | Leave a Comment »
Posted by naturalproductman on April 8, 2016
Martin Trmblay and co-workers from Infinity Pharmaceuticals have reported in Organic Process Research & Development on a scalable 6 to 7 membered ring expansion towards a hedgehog antagonist. When I looked at the structure it reminded me of cyclopamine.
ORPD paper
Posted in Cascade Reactions, Methodology, Ring expansion, Zinc | Leave a Comment »
Posted by naturalproductman on November 30, 2012
Kanchugarakoppal Rangappa and co-workers at the University of Mysore in India have reported in Tetrahedron Letters on the synthesis of thiophenes using a Simmons-Smith reagent.

TL paper
Posted in Cascade Reactions, Copper, Methodology, Named Reagents, Simmons-Smith, Transition Metal, Zinc | Leave a Comment »
Posted by naturalproductman on November 28, 2012
Bruce Lipshutz and co-workers have reported in JACS on a organocuprate Michael addition in WATER.

JACS paper
Posted in Cascade Reactions, Copper, Environmental, Methodology, Transition Metal, Zinc | Leave a Comment »
Posted by naturalproductman on August 22, 2012
Jin Kun Cha and co-workers at Wayne State University have reported in ACIE on a ring opening of cyclopropanol using stoichiometric diethyl zinc.

ACIE paper
Posted in Cascade Reactions, Methodology, Ring Opening, Sn2 prime, Transition Metal, Zinc | Leave a Comment »
Posted by naturalproductman on February 10, 2012
Tehshik Yoon and co-workers published in Organic Letters on a [2+2] photocycloaddition.

OL paper
Nessan Kerrigan and co-workers at Oakland University have reported in JACS on a [2+2] reaction between ketenes to form beta-lactones.

JACS paper
At first look at Daniel Romo’s name I thought of Tony Romo and funny enough they’re both in Texas. Here they report on a Mukaiyama Aldol lactonization to form beta-lactones.

JACS paper
Posted in Cascade Reactions, Cycloaddition, Light Mediated, Methodology, Mukaiyama, Named Reactions, Pericyclic reactions, Transition Metal, Zinc, [2+2] | Leave a Comment »
Posted by naturalproductman on December 5, 2011
Sang-gi Lee and co-workers have reported in Organic Letters on a synthesis of a substituted pyridine.

OL paper
Posted in Blaise, Cascade Reactions, Methodology, Named Reactions, Transition Metal, Zinc | Leave a Comment »
Posted by naturalproductman on September 18, 2011
Anthony Barrett and co-workers at Imperial College have reported in Organic Letters on a cascade reaction that involves the formation of a ketene followed by trapping with an enamine to form pyridinones.

OL paper
Posted in Cascade Reactions, Claisen condensation, Diels-Alder, Methodology, Named Reactions, Transition Metal, Zinc | Leave a Comment »
Posted by naturalproductman on August 18, 2011
Tamio Hayashi and co-workers at Kyoto University have reported in Chem Comm on a cobalt catalyzed Michael addition of an alkyne onto an enone.
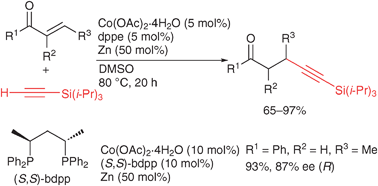
Chem Comm paper
Posted in Asymmetric, Cobalt, Methodology, Michael, Named Reactions, Transition Metal, Zinc | Leave a Comment »
Posted by naturalproductman on July 11, 2011
Matthew Sigman and colleague at the University of Utah have reported in JACS on an anti-Markovnikov addition of an alkyl group onto an alkene using an alkyl zinc reagent.

JACS paper
Posted in Cascade Reactions, Methodology, Palladium, Transition Metal, Zinc | Leave a Comment »