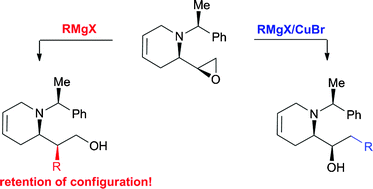
Pilar López-Ram-de-Víu and co-workers have reported in Organic and Biomolecular Chemistry on an epoxide opening reaction using a Grignard reagent – and the regioselectivity is switched when they use it in the presence of copper bromide.
Archive for the ‘Grignard’ Category
New Tricks with Grignard
Posted by naturalproductman on December 17, 2010
Chenzhong Cao and co-workers at Hunan University of Science and Technology have published in Organic Letters on a Grignard addition onto isopropenyl acetate to form 1,3-diols.

Posted in Cascade Reactions, Grignard, Methodology, Named Reactions | Leave a Comment »
Grignard Approach for Olefination
Posted by naturalproductman on April 22, 2010
Songlin Zhang and co-workers from the Chemical Engineering and Materials Science of Soochow University have recently reported in Organic and Biomolecular Chemistry on an olefination process that involved the addition of a Grignard reagent onto a ketone. The reaction forms an alkene in the presence of diethyl phosphite.

Posted in Cascade Reactions, Grignard, Methodology, Named Reactions | Leave a Comment »